Principles of an ITC Assay
What is Isothermal Titration Calorimetry?
Isothermal Titration Calorimetry (ITC) is an analytical technique with a basis in chemical thermodynamics that is used to study molecular interactions in solutions. The technique works by measuring enthalpy heat changes that occur during binding events between molecules, such as proteins, nucleic acids, and small molecules. Through direct measurement of the heat absorbed or released during these interactions, ITC titrations are able to provide valuable information regarding binding affinity, stoichiometry, and thermodynamic parameters of the binding interaction.
The ITC assay is fundamentally a stepwise titration of one species (e.g. a small molecule) into a calorimeter cell containing a solution of the other binding partner (e.g. a protein). The calorimeter is extremely sensitive, and any heat that is absorbed or released as a result of the binding interaction is measured and plotted on a thermogram, from which the enthalpy, stoichiometry, and binding affinity can be extracted.
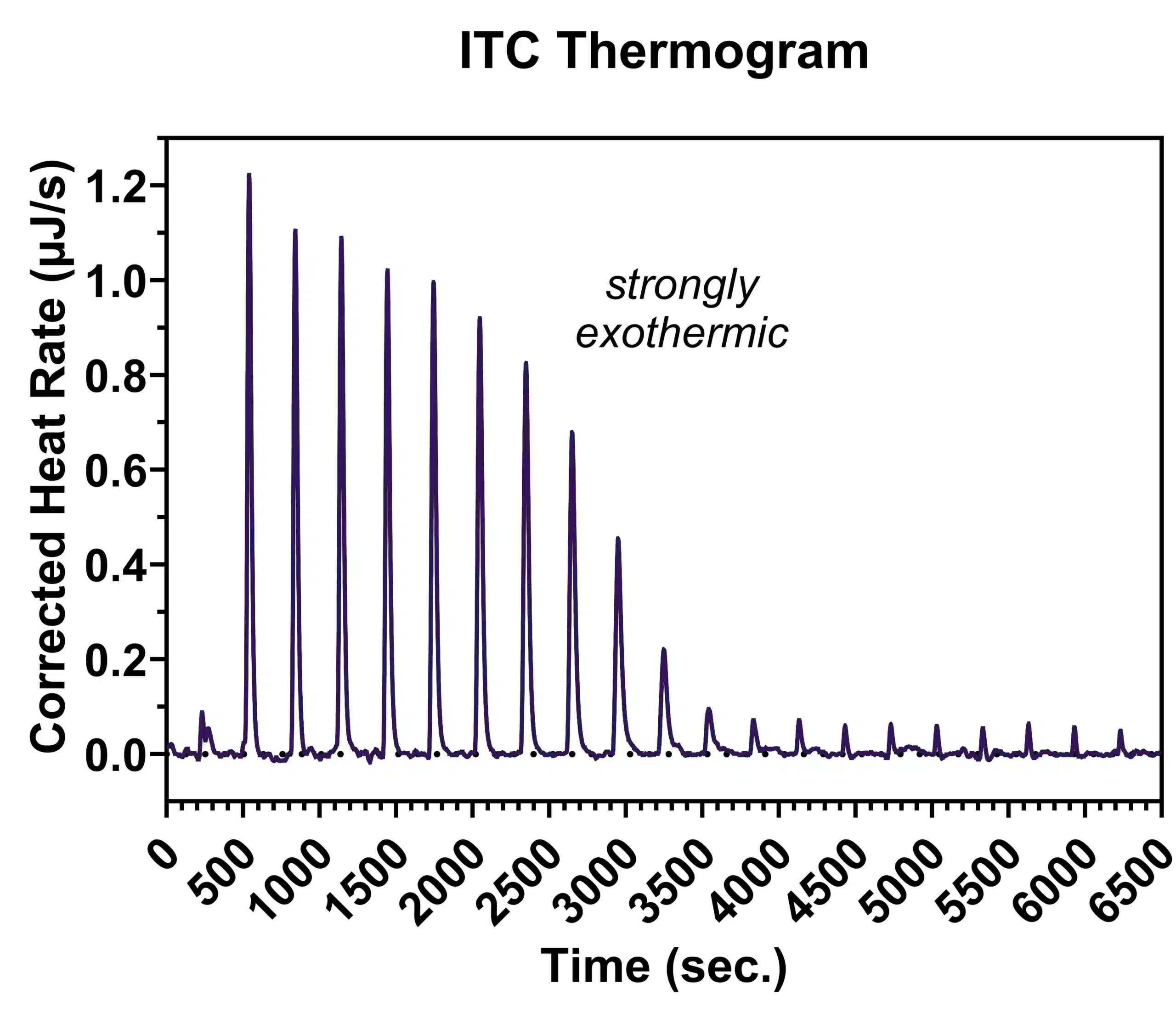
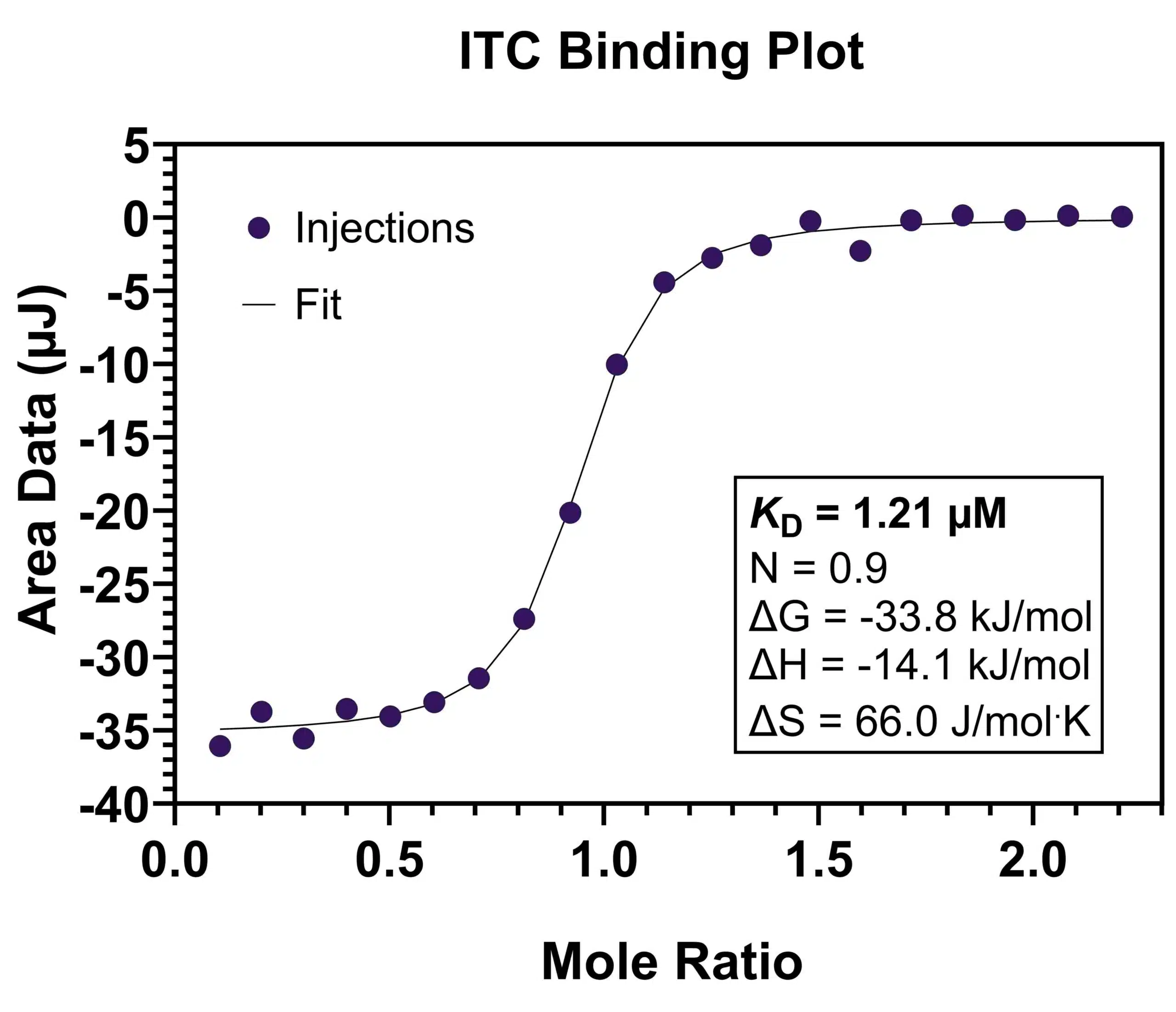
Application of ITC in Drug Discovery
An ITC binding assay is an indispensable tool in the drug discovery toolkit. While it is a lower throughput technique compared to SPR, it is a truly solution phase approach that is often needed as an orthogonal binding confirmation or in cases where surface-based techniques fail. ITC provides numerous pieces of quantitative data that are used to advance drug discovery and development.
Binding affinity: Binding affinity is a quantification of the degree to which a ligand binds to its target and is represented in terms of concentration (molarity). ITC enables native binding affinities to be determined, allowing for a direct comparison and optimization of potential drug candidates. ITC is effective in quantifying binding affinity to the low nanomolar range.
Stoichiometry: Stoichiometry refers to the binding ratio between the ligand and the target macromolecule (i.e. 1:1, 2:1, etc.).
Thermodynamic parameters: Through ITC, data on the thermodynamic aspects of molecular interaction can be determined, such as information regarding enthalpy (i.e. whether an interaction is exothermic or endothermic) and entropy.
Influence of assay factors: ITC assays can inform on the effects of variables such as temperature, buffer composition, and pH.
Our Advanced ITC Assay Services
ITC is one of the most preferred methods for studying molecular interactions, as it offers several advantages over other biophysical techniques.
Label-free detection: ITC requires no labels, tags, fluorophores, or surface immobilization; it is a truly solution-phase biophysical technique. By preserving the native state of the biomolecules, ITC provides an accurate representation of their interaction under physiological conditions.
Quantitative Analysis: ITC provides quantitative data for binding affinity, enthalpy, and, stoichiometry, enabling precise characterization and comparison of molecular interactions.
Broad Applicability: ITC is a highly versatile technique. It can analyze protein-ligand, protein-protein, and nucleic acid-ligand interactions, among others, proving to be an invaluable tool for researchers across a variety of disciplines.
Sample Requirements and Throughput: The main shortcoming of ITC revolves around sample consumption and throughput, which is why we recommend it as a secondary screening measure to techniques such as SPR. Our TA Instruments Nano ITC requires less sample than other ITC instruments, both contemporary and historical, requiring approximately 200-300 µL of sample for the analysis cell. We can typically acquire 3-5 full, high-quality titrations per 8 h analysis period.
Further Information on ITC
Competitive Binding Experiments
Overview of the design and execution of competitive inhibitor experiments via ITC, with particular relevance to fragment screening.
Obtaining Enzyme Kinetics with ITC
Application Note for using ITC to obtain enzyme kinetic constants.
Related Products & Services:
Although ITC assay is an excellent secondary or orthogonal screen for hit validation and characterization, another technique such as SPR (Surface Plasmon Resonance) is recommended for primary and high-throughput screening.

High-Throughput Primary Screening via SPR

We offer SPR services for high-throughput fragment screening and hit validation, including fragment clean screens, binding level screens, and titration for binding confirmation and kinetics.
