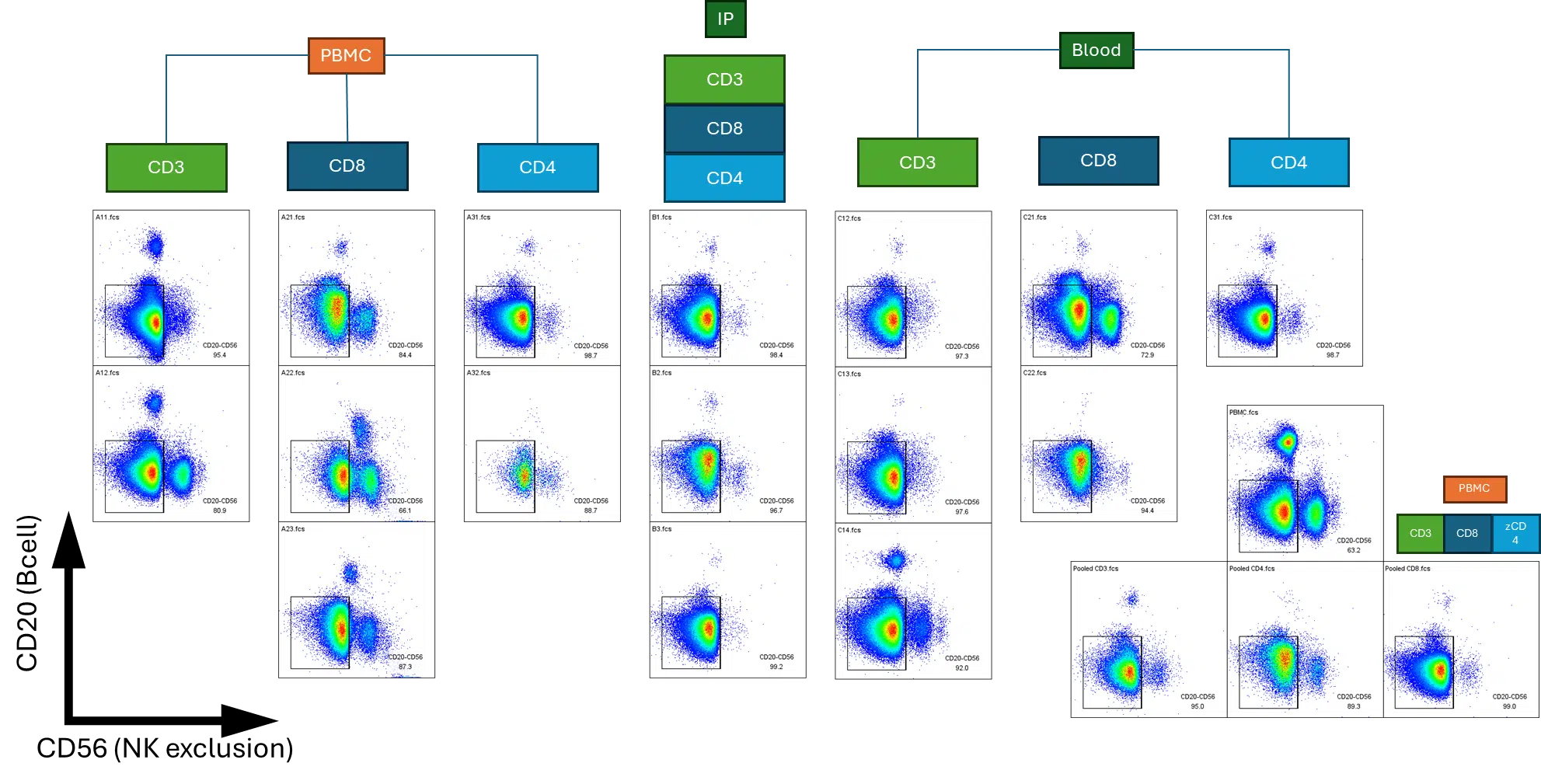
Cell therapy success starts with the right cells. We isolate and define primary human T cell subsets from multiple biological sources with rigorous control over purity, viability, and phenotype. We routinely achieve greater than 80% purity in isolated CD3+, CD4+, and CD8+ T cell populations.
Featured Capabilities
Comprehensive control of primary T cell isolation, activation, and functional performance to support cell therapy development.
PBMC-to-T Cell Precision
Our capabilities extend from blood fractionation for serum, plasma, and diverse cell populations to fully customized PBMC processing and phenotypic characterization workflows.
Controlled activation states
Activation is executed under standardized stimulation conditions and confirmed through comprehensive marker-based validation.
Engineering-ready cells
Activation and expansion conditions optimized to enhance viral transduction efficiency, gene editing performance, and downstream construct validation.
Donor & Source Flexibility
We work with a variety of donors to adapt to complex study designs with attention to immunophenotype and HLA considerations when relevant to program objectives.
Controlled Primary T Cell Isolation Across Variable Sources

Isolation establishes input quality. Activation defines functional potential. Together, they demonstrate something more powerful: control over the biological system itself. Our activation workflows produce reproducible and tunable shifts in T cell phenotype, enabling controlled expansion of defined functional subsets. By modulating stimulation conditions, we direct naïve, memory, and effector distributions with consistent performance across donors. This level of biological control strengthens transduction efficiency, improves potency modeling, and supports the persistence and scalability required for successful cell therapy development.
Frequently Asked Questions
Answers to some of the most common questions about preclinical ophthalmic research service offerings.