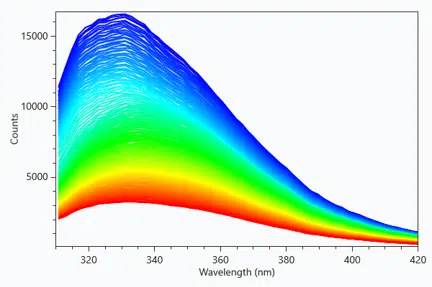
NanoDSF is a type of Differential Scanning Fluorimetry used to determine conformational protein stability by employing intrinsic tryptophan or tyrosine fluorescence, as opposed to the use of extrinsic fluorogenic dyes that can perturb the system and affect protein stability.
We exclusively use the SUPR-DSF spectrometer from Protein Stable for our DSF measurements.
Reliably measure protein Tm values
The SUPR-DSF instrument measures protein conformational stability via thermal denaturation (10 oC - 105 oC) or chemical denaturation (chaotropes).
Label-Free
nanoDSF utilizes intrinsic protein fluorescence, meaning there’s no need for adding dyes.
Native Sample Measurement
Samples can be analyzed as-is without dilution, rebuffering, or the addition of exogenous fluorophores.
Minuscule Sample Requirements
10 µL of sample is all that’s needed. Sample concentrations as low as 0.1 mg/mL.
Full Spectrum Data
Fluorescence data is recorded from 310nm-420nm for every sample, enabling global data fitting.
High-Throughput
Measure 384 samples or replicates per experiment. We pass the value and cost savings onto you.
What sets Ichor’s nanoDSF services apart?
Ichor’s nanoDSF services include sample handling, standard assay execution, data analysis & reporting, and development of custom workflows for unique projects.
Small Molecule Binding Studies
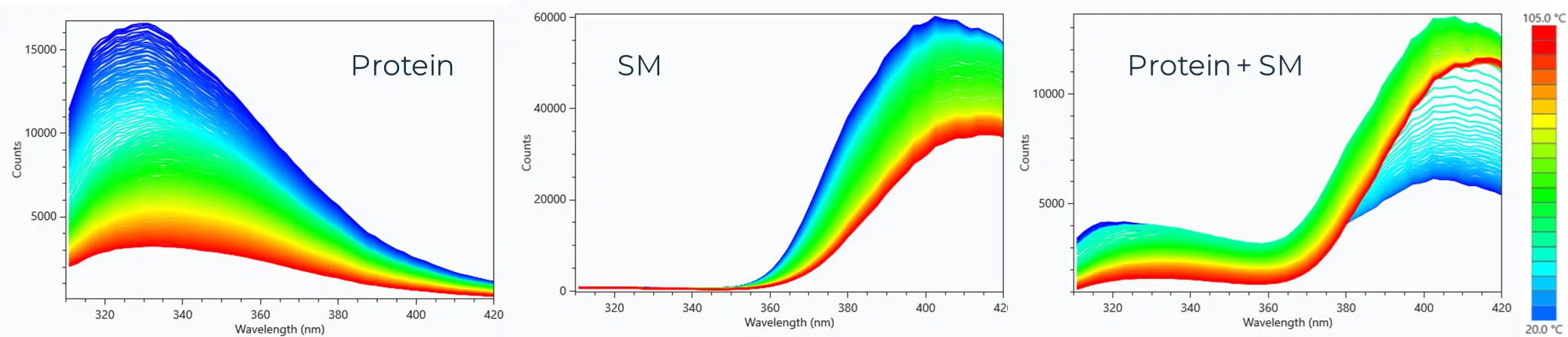
Ichor’s nanoDSF services go beyond routine formulation screening experiments and into the realm of small molecule binding studies. Traditional DSF assays are very limited in their tolerance of small molecules due to the constraint of only measuring two emission intensities at 330 nm and 350 nm. If a small molecule has an autofluorescent contribution to these two filter sets, an accurate shift in the Tm will not be measurable. The SUPR-DSF uses a CMOS fluorimeter which gives us emission intensity between 310 nm and 420 nm, making it possible to choose a range that is free of interference.
Data Analysis with the SUPR-DSF
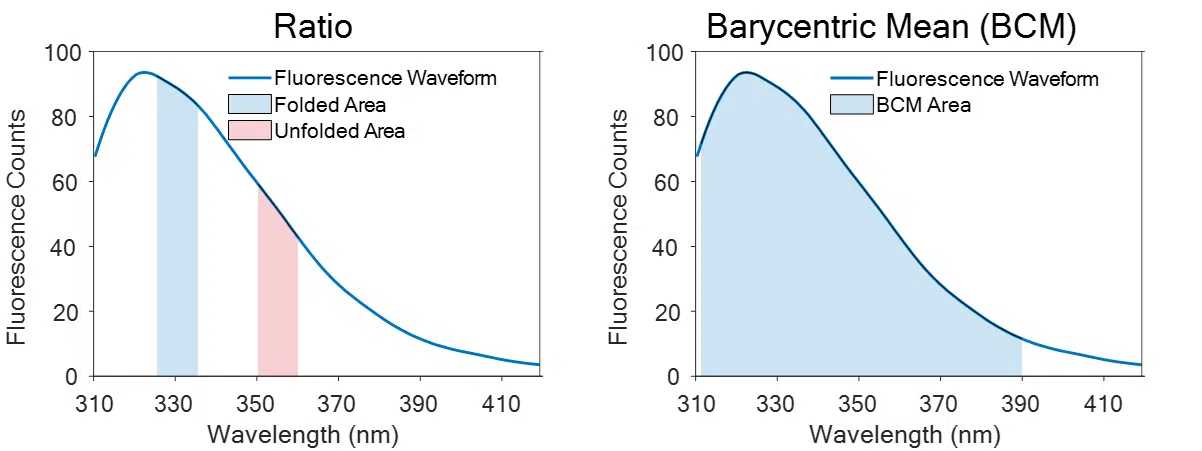

Barycentric Mean Fluorescence Analysis
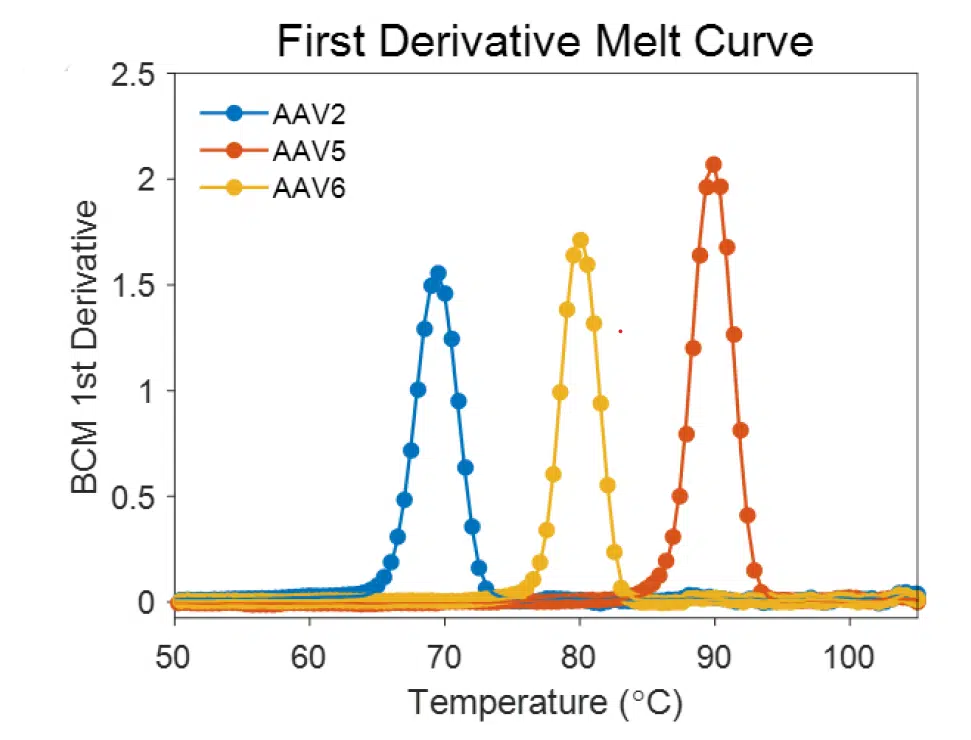
Barycentric Mean: BCM measures the center of mass of the fluorescence waveform, enabling the computation of wavelength shifts as the protein unfolds. BCM produces better results than the ratio method by taking advantage of the entire fluorescence spectrum.
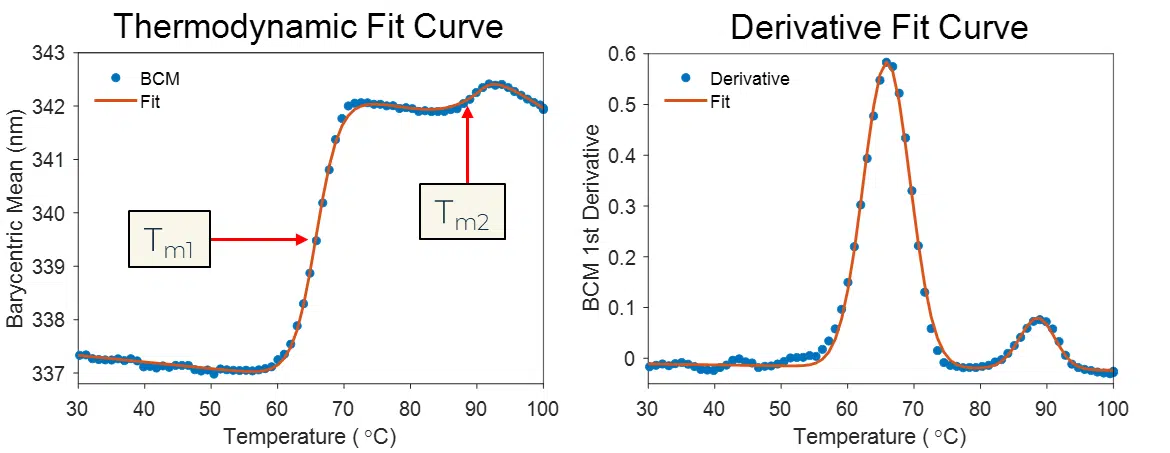

Melt Curve Analysis
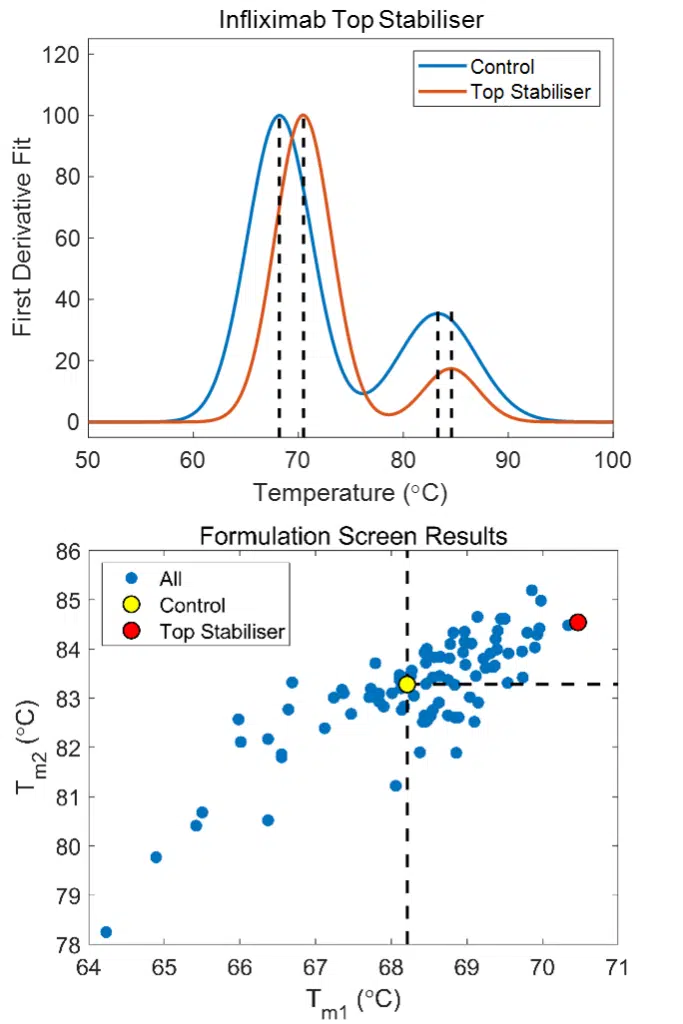
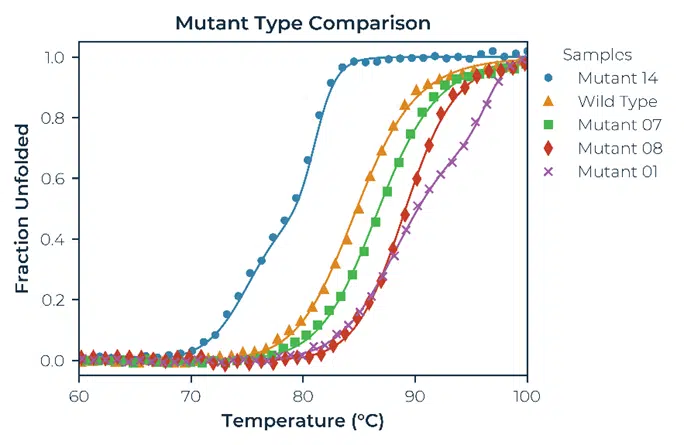
The data that results from the BCM fluorescence analysis can be fit using either a thermodynamic model or by taking the 1st Derivative and calculating the inflection points.
Featured Capabilities / Case Studies – Courtesy of Protein Stable
Frequently Asked Questions
Learn more about Ichor’s nanoDSF services by reading below.