Case Study: Establishing Novel FRET Protocols with Novel Tagging Systems
August 5, 2021
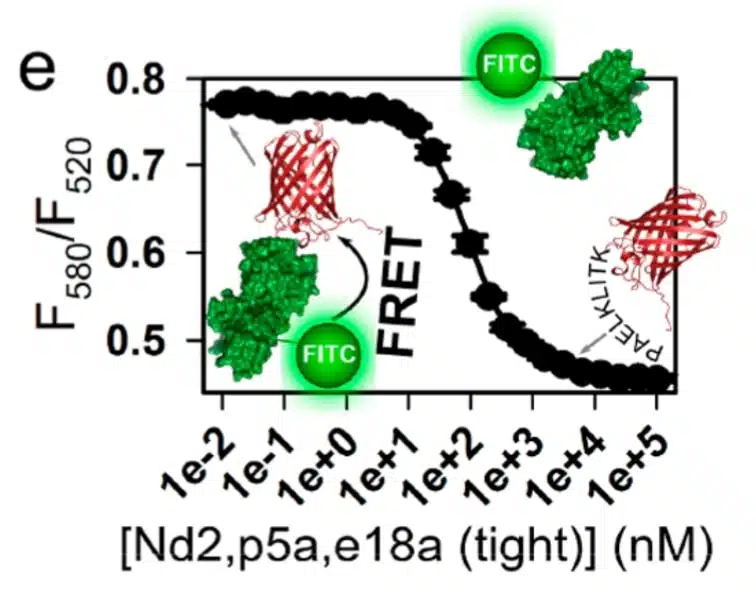
In 2018, we published an article highlighting the creation of a generalizable protein binding scaffold and epitope tag that is orally bioavailable called RPtag. RPtag is based on a ribose-binding protein isolated from an extremophile, awarding it expansive stability across highly variable environmental conditions. It was rationally engineered by splitting an irregular C-terminal pair of beta sheets, generating large and small fragments with high affinity and specificity to each other. Like antibodies, RPtag binds tightly to its epitope, but unlike antibodies, RPtag is small (23kD), allowing it’s use where antibodies fail. In developing RPtag, we established new protocols in fluorescence resonance energy transfer (FRET) assay techniques. Our protocol promotes consistent quantitative analysis of interactions between molecules with nanometer precision.
At Ichor Discovery, knowing what questions to ask and acknowledging the limitations of commonly understood techniques endows us a perspective that perpetuates the creation of novel solutions to problems we run into. We thrive on progress, and our ability to devise methods in-house with unparalleled efficiency is an asset for any project.
Biochemistry 2018, 57, 21, 3036–3049
Publication Date: May 3, 2018 https://doi.org/10.1021/acs.biochem.8b00170