Case Study: Creation of Novel Biophysics-Based Protocols
August 5, 2021
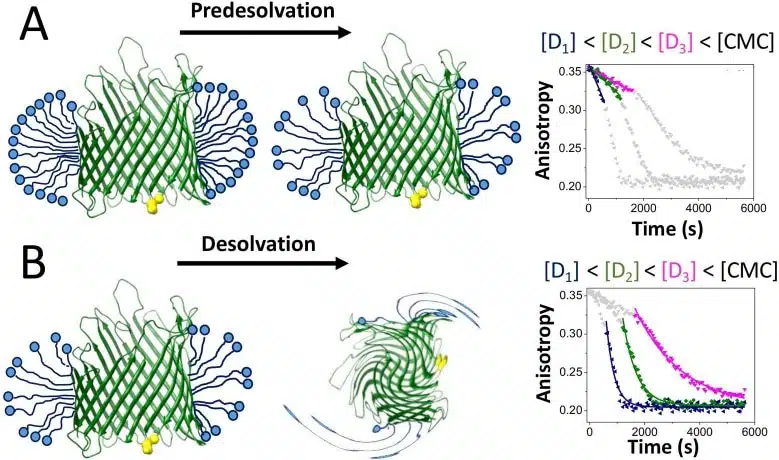
In 2020, we published an article titled “High Throughput Screening of Protein-Detergent Complexes Using Fluorescence Polarization Spectroscopy”, where we outlined novel protocols to uncover the mechanisms of membrane protein and solubilizing detergent interactions. Our protocol uses steady-state fluorescence polarization spectroscopy to observe the rotational mobility of proteomicelle complexes, the formation generated by the binding of membrane proteins with detergent monomers. Labeled membrane proteins and detergents form protein-detergent complexes (PDC) that demonstrate a slowed rotational diffusion with respect to the unbound protein, decreasing the emission in perpendicular planes and increasing emissions in parallel planes to the polarized light. We are able to determine the amount of bound proteins in solution according to this change in emission by analyzing steady state FP anisotropy relative to the equilibrium dissociation constant, and can be used with any membrane protein that forms complexes with detergent micelles.
This is an example of an innovational approaches born out of our problem agnostic approach, allowing us to adapt existing and produce novel techniques to answer questions previously unanswered. In addition to detergent-protein interactions, our philosophy has been applied to research on production analysis of intrinsically disordered proteins and multi-unit protein complexes, both traditionally difficult problems in biophysics.
Protein Science, 97, e96. doi: 10.1002/cpps.96