Our Toxicology Services
Acute Toxicity Studies
Establish the maximum tolerated dose (MTD) level of your test compound in a novel species, as well as identify potential adverse effects and determine dose levels for future studies.
Dose-Range Finding Studies
Dose-range finding (DRF) studies are important to conduct prior to the definitive GLP studies to assess potential adverse effects and determine tolerable dose levels. We offer single and repeat-dose designs.
Carcinogenicity Studies
Discover the potential tumorigenicity of your long-term use compound. Ichor has extensive experience in both life-span studies and carcinogenic models, creating the perfect blend of knowledge and skills.
Chronic/Subchronic Toxicity
With Ichor’s team of life-span study experts, we are ready to provide you with comprehensive data to inform decisions regarding the safety of your test compound after long-term exposure in subchronic and chronic studies.
Ocular Toxicity
Let our team of specialists demonstrate why we’re one of the fastest growing preferred partners in ophthalmology! With capabilities in ocular dose administration and a wide variety of techniques, we’re ready to deliver high quality results.
Routes of Administration
- Oral Gavage
- Intraperitoneal
- Intravenous (bolus, slow push, retro-orbital)
- Intramuscular
- Subcutaneous
- Ocular (intravitreal, subretinal, intracameral, and suprachoroidal)
- Topical/Dermal
- Inhalation
- Intracranial ventricular
- Intranasal
- Dietary
- Vaginal/Anal
Investigative Studies

Our preclinical toxicology research team is ready to help you further investigate the tolerability and toxicity of your compound. By partnering with Ichor Life Sciences, you’re choosing a capable and dedicated team to help with your investigative and nonGLP studies allowing you to enter larger-scale projects with confidence.
Model Highlight
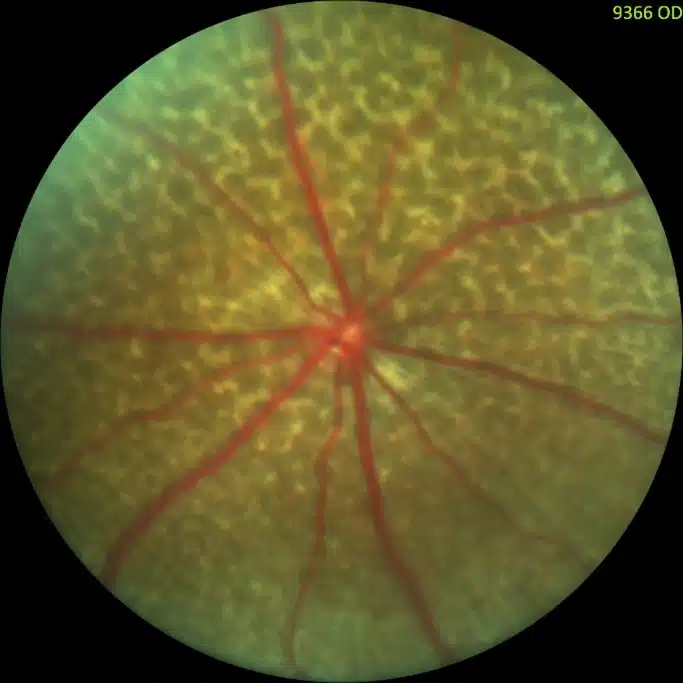
Our team is quickly becoming the most preferred partner for ocular studies. We offer a variety of validated preclinical models, ocular PK/PD services, and utilize the industry-best equipment to conduct our studies. We offer tonometry (IOP) evaluation, electroretinography (ERG), enhanced microscopic evaluation, ocular histopathology, ocular and systemic tissue distribution services, and more.
Capabilities and Services Summary
Frequently Asked Questions
Answers to some of the most common questions about preclinical toxicity research.


